Abstract
Phakic intraocular lens implantation (Phakic APL) is an established and effective refractive solution for moderate to high myopia and other refractive errors, particularly in patients with thin corneas or unsuitable corneal biomechanics for laser refractive procedures. Accurate sizing of Phakic (APL) lenses is essential to achieve optimal postoperative vault, defined as the distance between the posterior surface of the phakic lens and the anterior surface of the crystalline lens. An appropriate vault ensures long-term safety, stable visual outcomes, and minimal risk of complications such as cataract formation or anterior chamber angle compromise.
Traditional sizing methods rely on external anatomical parameters such as White-to-White (WTW) and Anterior Chamber Depth (ACD), which provide indirect estimation of internal ocular anatomy. Recent advances in anterior segment imaging technologies, such as Optical Coherence Tomography (OCT), Ultrasound Biomicroscopy (UBM), Scheimpflug tomography, and Artificial Intelligence (AI)-based multivariable prediction models, enable improved anatomical characterisation of internal ocular structures, including Angle-to-Angle (ATA), Sulcus-to-Sulcus (STS), and Lens Rise. These modern approaches enhance the predictability of vault outcomes and support personalised phakic lens sizing strategies.
1. Anatomical Basis of Phakic intraocular lens (APL) Positioning
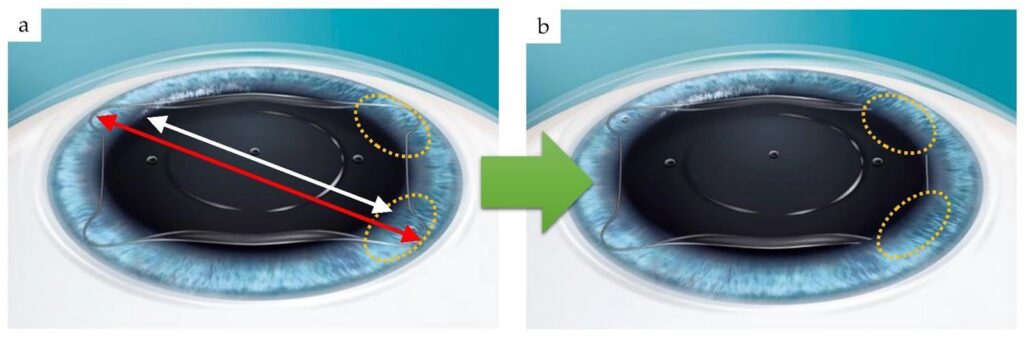
Phakic (APL) lenses are positioned in the posterior chamber, where the haptics rest in the ciliary sulcus behind the iris and in front of the crystalline lens. The size of the lens must correspond closely to the anatomical dimensions of this internal structure to achieve stable positioning and appropriate vault.
Key anatomical factors influencing Phakic (APL) sizing:• corneal diameter
• anterior chamber depth
• ciliary sulcus diameter
• iris contour
• crystalline lens position
• angle configuration
Because the ciliary sulcus cannot always be directly visualised using conventional external measurements, multiple surrogate and direct imaging parameters are used to estimate appropriate lens size.
Vault is defined as:
The distance between the posterior surface of Phakic (APL) and the anterior crystalline lens.
Typical clinical interpretation:
| vault range | interpretation |
|---|---|
| <200 µm | low vault |
| 300–700 µm | optimal |
| 700–1000 µm | acceptable |
| >1000 µm | high vault |
Vault reflects compatibility between lens size and internal ocular anatomy
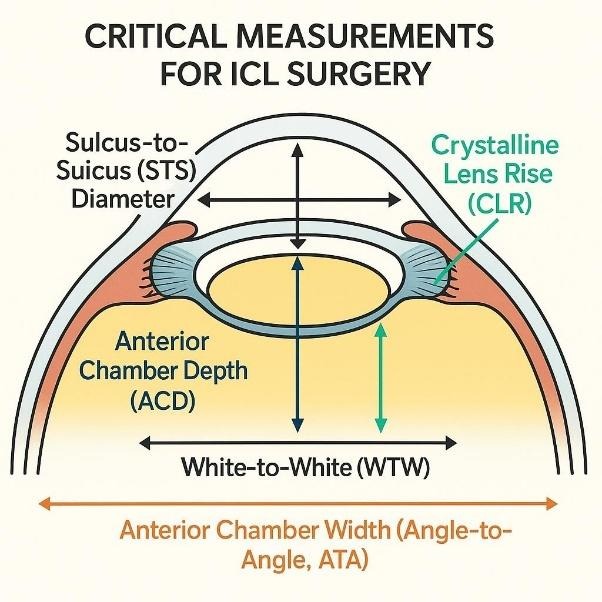
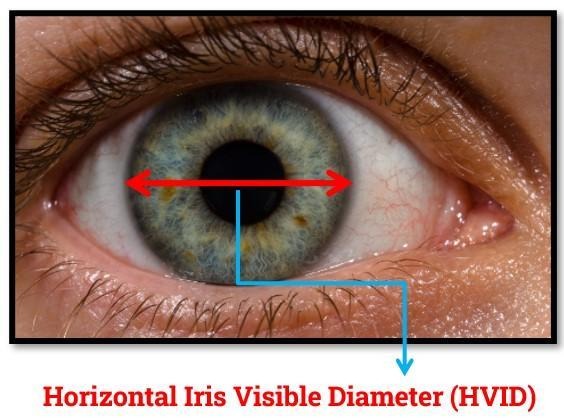
WTW measures the horizontal corneal diameter between the nasal and temporal limbal boundaries and is commonly used as a primary parameter in ICL sizing due to its simplicity and accessibility. The main advantages of WTW include ease of measurement, non-contact acquisition, and widespread availability across most optical biometers and corneal imaging systems. However, an important limitation of WTW is that it represents an external anatomical dimension and does not directly correspond to the internal ciliary sulcus diameter, where the ICL haptics are positioned. Consequently, reliance on WTW alone may not fully capture individual variations in internal ocular anatomy that influence postoperative vault.
Anterior Chamber Depth (ACD) represents the distance between the posterior corneal surface (endothelium) and the anterior surface of the crystalline lens. ACD reflects the available internal space in the anterior segment for placement of the Phakic (APL) lens.
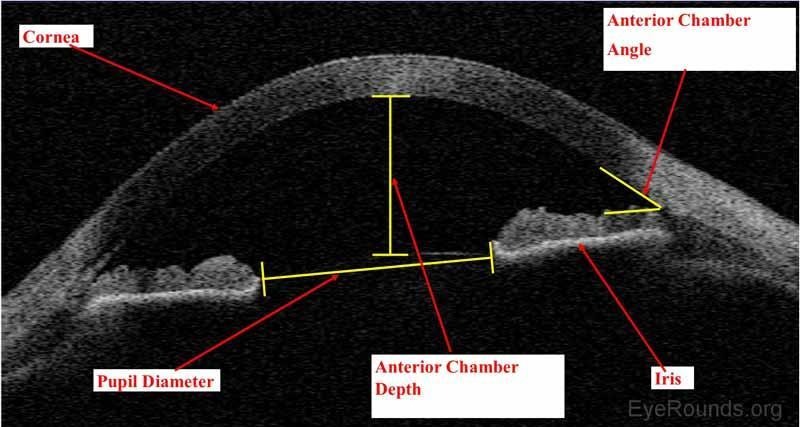
ACD is influenced by:
A deeper anterior chamber typically provides greater internal volume for lens positioning and may influence vault behaviour.
The traditional combination of white-to-white (WTW) and anterior chamber depth (ACD) may not adequately represent the complex variability of internal anterior segment anatomy. Since these parameters primarily reflect external or indirect anatomical dimensions, they may fail to capture individual differences in sulcus size, lens rise, and ciliary body configuration. As a result, clinical outcomes may demonstrate variability in postoperative vault, including cases of unexpectedly high vault or unexpectedly low vault. Additionally, noticeable inter-eye variability may occur even when preoperative biometric measurements appear similar, highlighting the limitations of relying solely on WTW and ACD for accurate ICL sizing.
4.1.1. Definition and Anatomical Concept
Angle-to-Angle (ATA) represents the internal horizontal distance between the scleral spurs across the anterior chamber. The scleral spur is an anatomical landmark located at the junction between the cornea and sclera, forming part of the anterior chamber angle structure.
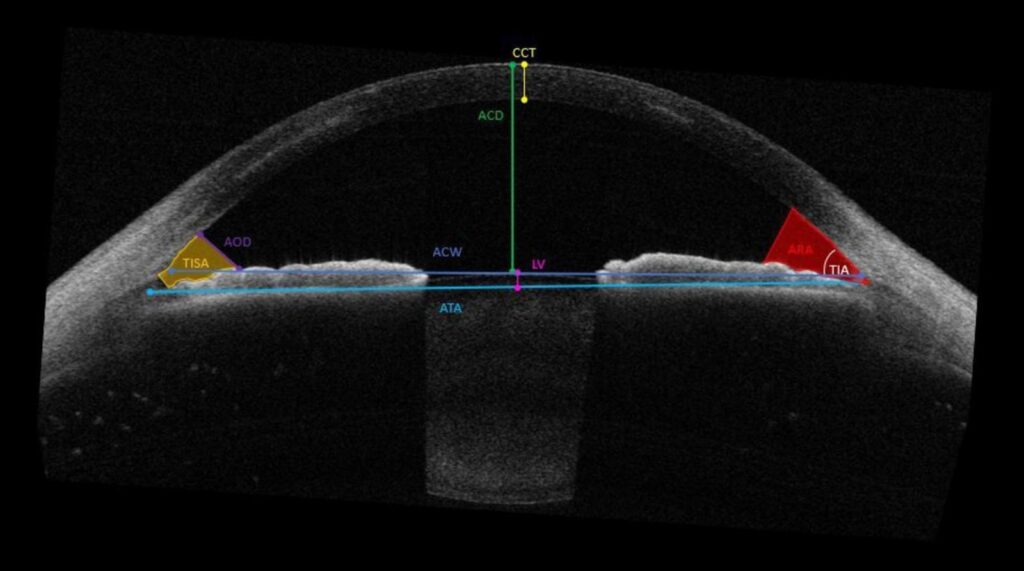
ATA measurement provides a more anatomically relevant estimation of internal ocular width compared with WTW because it evaluates internal structures closer to the anatomical location where Phakic (APL) lenses interact with surrounding tissues.
While WTW measures the external corneal diameter, ATA reflects the internal structural width of the anterior chamber, providing additional information regarding spatial geometry of the anterior segment.
4.1.2. Measurement Method
ATA is typically measured using:
These devices identify scleral spur landmarks across multiple meridians and calculate the internal anterior chamber diameter.
Clinical Importance in Phakic (APL) Sizing
ATA provides an improved anatomical representation of the internal ocular dimension compared with external corneal measurements. Differences between WTW and ATA values may indicate variation in scleral thickness, angle configuration, or anterior segment morphology.
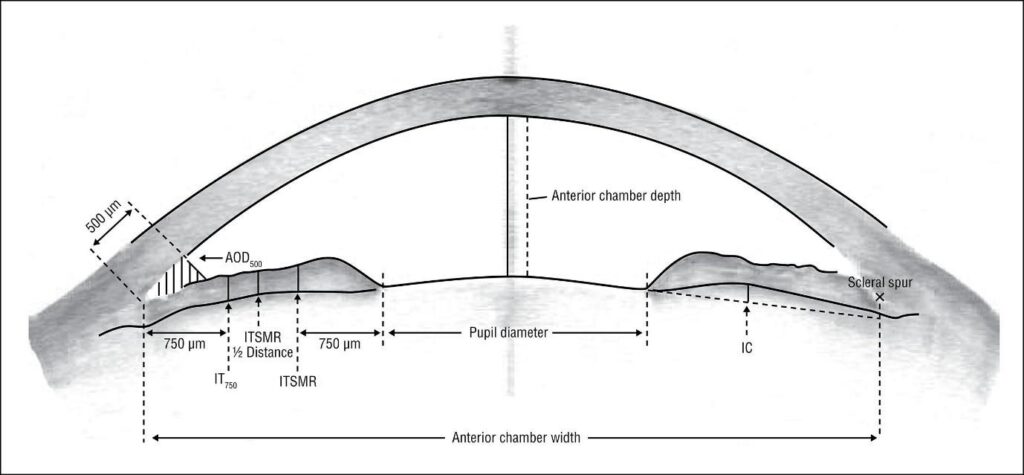
ATA is particularly useful in cases where:
WTW values appear borderline
External measurements show inter-eye symmetry, but the vault differs
Anterior chamber geometry appears atypical
ATA measurement helps refine the estimation of internal ocular width and may contribute to improved lens size selection.
4.1.3. Relationship between ATA and Vault
: A larger ATA may indicate a larger internal anatomical width and may be associated with a lower vault if lens size is relatively small.
A smaller ATA may indicate narrower internal anatomy and may be associated with a higher vault when lens size is relatively large.
ATA improves understanding of spatial anatomy, influencing vault behaviour.
4.2.1. Definition and Anatomical Concept
Sulcus-to-Sulcus (STS) represents the internal diameter between opposing ciliary sulcus structures, corresponding directly to the anatomical location where Phakic (APL) haptics rest.
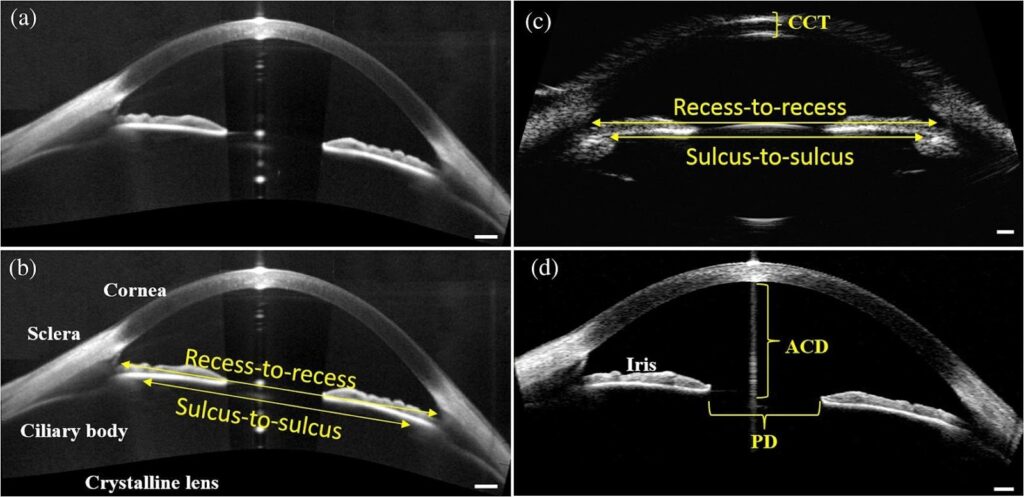
The ciliary sulcus is located posterior to the iris root and anterior to the ciliary body. Because the Phakic (APL) lens is supported within this anatomical space, STS measurement provides a direct representation of the anatomical dimension most relevant to lens sizing.
4.2.3. Measurement Method
STS is typically measured using:
Ultrasound Biomicroscopy (UBM)
high-resolution anterior segment imaging techniques capable of visualizing posterior iris structures
UBM imaging allows visualisation of the ciliary body and sulcus morphology, enabling measurement of the internal diameter relevant to lens positioning.
4.2.4. Clinical Importance in Phakic (APL) Sizing
STS is considered one of the most anatomically relevant parameters for predicting postoperative vault because it directly represents the internal support structure of the lens.
Variation in STS diameter explains why eyes with similar WTW values may demonstrate different vault outcomes.
STS measurement is particularly valuable in cases demonstrating:
4.2.5. Relationship between STS and Vault
: A larger STS diameter may result in a lower vault if the lens size is relatively small.
A smaller STS diameter may result in a higher vault if the lens size is relatively large.
Direct measurement of sulcus anatomy improves the predictability of effective lens fit.
4.3.1 Definition and Anatomical Concept
Lens rise represents the perpendicular distance between the anterior surface of the crystalline lens and a reference line connecting the scleral spurs (angle plane).
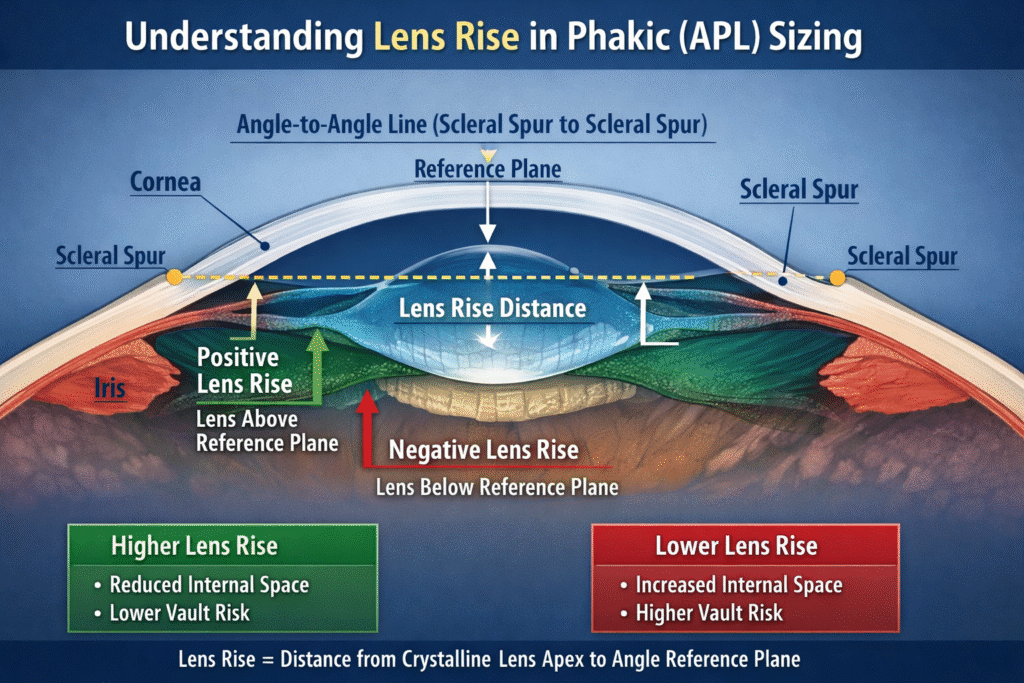
Lens rise describes the relative anterior positioning of the crystalline lens within the eye and provides information about how much the crystalline lens protrudes into the anterior chamber space.
Lens rise may be positive or negative, depending on the relative position of the crystalline lens apex with respect to the angle reference plane.
4.3.2. Measurement Method
Lens rise is measured using:
These devices identify the scleral spur plane and calculate the relative position of the crystalline lens apex.
4.3.3. Clinical Importance in Phakic (APL) Sizing
Lens rise influences the effective space available between the crystalline lens and phakic lens. Eyes with higher lens rise demonstrate reduced internal space, which may influence postoperative vault behaviour even when WTW and ATA values appear similar.
Lens rise is particularly important in cases where:
ACD appears adequate, but vault prediction remains uncertain
Age-related lens thickening occurs
An unexpectedly low vault is observed despite an appropriate lens size
Relationship between Lens Rise and Vault
higher lens rise → reduced effective internal space → tendency toward lower vault
lower lens rise → increased internal space → tendency toward higher vault
Lens rise helps explain variability in vault outcomes between eyes with similar external dimensions.
Anterior Segment Optical Coherence Tomography (AS-OCT) is one of the most important modern diagnostic technologies used in Phakic (APL) sizing. OCT uses low-coherence interferometry to generate high-resolution cross-sectional images of the anterior segment, allowing detailed visualisation of anatomical structures relevant to lens sizing.
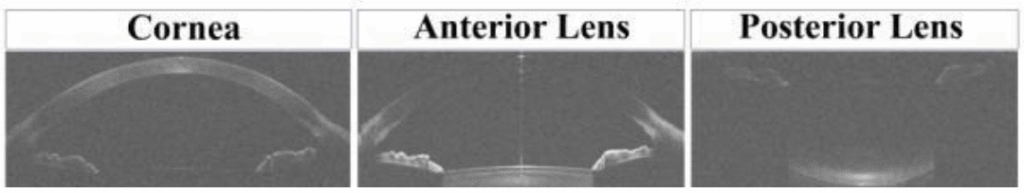
Key anatomical parameters measured using AS-OCT
Angle-to-Angle (ATA)
The distance between scleral spurs across the anterior chamber.
ATA provides better internal anatomical estimation compared to WTW.
Anterior Chamber Depth (ACD)
distance from the endothelium to the crystalline lens.
determines available internal space for lens placement.
Lens Rise
The distance between the crystalline lens apex and angle reference plane.
important determinant of vault behaviour.
Angle anatomy
evaluation of angle width and configuration.
helps predict the risk of angle crowding in high vault cases.
Advantages of Phakic (APL) sizing
non-contact measurement
high repeatability
high spatial resolution
visualisation of internal anatomy
good patient comfort
These devices provide detailed anatomical mapping useful for personalised lens sizing.
5.2 Ultrasound Biomicroscopy (UBM)
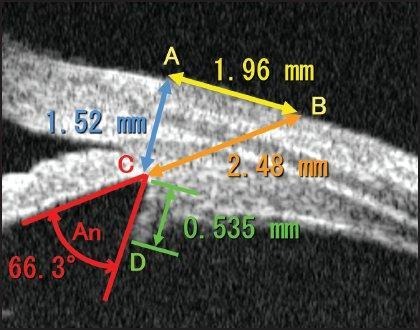
Ultrasound biomicroscopy utilises high-frequency ultrasound waves (35–50 MHz) to image structures posterior to the iris that are not easily visualised by optical methods.
UBM remains one of the most anatomically direct methods for evaluating internal ocular structures relevant to Phakic (APL) sizing.
Anatomical parameters obtained using UBM
Sulcus-to-Sulcus (STS)
direct measurement of ciliary sulcus diameter.
represents the anatomical resting location of Phakic (APL) haptics.
Ciliary body configuration
The shape and orientation of ciliary processes influence lens positioning.
Iris insertion profile
affects lens centration and vault stability.
Posterior chamber morphology
helps identify anatomical variations influencing the vault.
Clinical significance
STS measurement provides a direct anatomical reference for lens sizing.
particularly useful when:
WTW borderline
Unexpected vault previously observed
high myopia cases
Anatomical asymmetry suspected
Advantages
direct visualisation of sulcus anatomy
improved anatomical accuracy
useful in complex cases
Limitations
contact or immersion technique required
operator dependent
limited availability in routine clinical settings
5.3 Scheimpflug Imaging (Corneal Tomography)
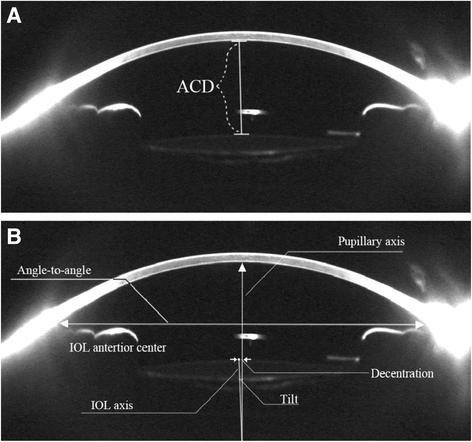
Scheimpflug imaging systems use a rotating camera and slit illumination to create a three-dimensional reconstruction of the anterior segment.
Scheimpflug tomography is primarily used for corneal assessment, but also provides useful parameters for Phakic (APL) sizing.
Parameters relevant to Phakic sizing
Optical biometers use infrared interferometry to measure ocular dimensions.
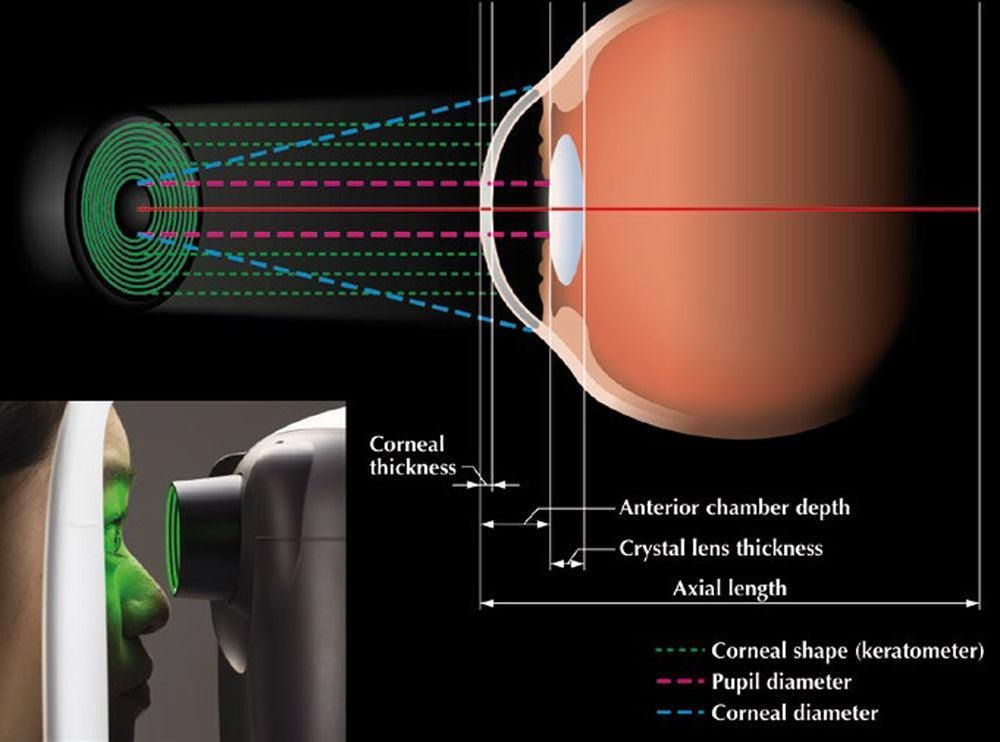
Although originally developed for cataract surgery planning, optical biometers provide useful parameters for Phakic (APL) sizing.
Parameters obtained
Clinical relevance
: optical biometry provides standardised measurement of WTW and ACD with high repeatability.
often used as a baseline measurement before advanced imaging.
Artificial Intelligence (AI) is increasingly transforming the methodology of Phakic (APL) sizing by enabling integration of multiple anatomical and biometric parameters into predictive models capable of estimating postoperative vault with improved precision. Traditional sizing approaches rely primarily on limited variables such as WTW and ACD, whereas AI-based systems analyse complex, nonlinear relationships between multiple anatomical factors that influence lens positioning within the posterior chamber.
Modern AI algorithms utilise machine learning (ML), deep learning, and multivariable regression models trained on large clinical datasets containing preoperative biometric parameters and postoperative vault outcomes. These models identify patterns between internal ocular anatomy and vault behaviour that may not be evident using conventional nomograms. AI-based prediction has demonstrated improved performance in predicting postoperative vault and optimising lens size selection compared with traditional estimation methods.
Several AI-assisted platforms and research-based tools are currently available or emerging in clinical practice for Phakic (APL) sizing:
6.1.2. ICL Guru (RevAI platform)
One of the most advanced AI-driven vault prediction systems uses deep learning models trained on ultrasound biomicroscopy (UBM) image datasets to predict postoperative vault behaviour. The system analyses anatomical parameters, including internal anterior segment geometry, anterior chamber depth, and sulcus configuration, to generate a predicted vault range for different lens sizes.
A key feature of this approach is the ability to analyse multiple image frames from UBM scans, automatically identify optimal anatomical landmarks, and simulate expected vault outcomes. AI-based frame selection improves measurement consistency and reduces operator dependency.
6.2. Machine Learning Vault Prediction Models
Recent studies have demonstrated the use of machine learning techniques such as:
• Random forest models
• Gradient boosting (XGBoost)
• multivariable regression algorithms
• neural networks
These models utilise combinations of anatomical parameters, including WTW, ATA, STS, lens rise, keratometry, and age, to predict vault outcomes more accurately than traditional formulas.
Machine learning approaches have shown improved accuracy in predicting vault variability, especially in anatomically borderline eyes.
6.3 Multimodal AI-based Biometric Integration
Modern AI tools increasingly integrate data from multiple imaging modalities:
This multimodal integration improves prediction reliability by combining structural and biometric information into a unified analytical model.
AI models can simultaneously analyse:
Such multivariable integration allows more individualised prediction of vault behaviour.
Step 1
input biometric data including WTW, ACD, ATA, STS, keratometry and refraction.
Step 2
An AI algorithm compares input parameters with a database of previously implanted cases.
Step 3
The predictive model estimates the vault range for each available lens diameter.
Step 4
The clinician selects the lens size corresponding to the optimal predicted vault range.
Some platforms also provide confidence intervals or safety bands indicating the probability of achieving the target vault range.
AI-based systems provide several clinical advantages compared with traditional sizing approaches:
AI models can identify subtle anatomical relationships that influence vault behaviour but may not be evident from isolated measurements.
AI-based systems are particularly beneficial in cases with:
Despite promising results, AI-based sizing systems face certain limitations:
dependence on the quality of imaging data
variability in device measurement standards
limited availability of large standardised datasets
regulatory and validation requirements
Further multicentre validation studies are required to ensure universal applicability across diverse populations.
AI-assisted Phakic (APL) sizing represents an evolution from empirical estimation toward data-driven personalised refractive surgery planning. Integration of AI with advanced imaging technologies enables a more comprehensive understanding of internal ocular anatomy and supports improved consistency in vault outcomes.
As datasets continue to expand and predictive algorithms improve, AI-based systems are expected to play an increasingly important role in enhancing safety, predictability, and clinical confidence in Phakic (APL) sizing workflows.
8. 3D Anterior Segment Modelling for Phakic (APL) Sizing
What it is, availability, and role in ICL sizing
What is 3D Anterior Segment Modelling?
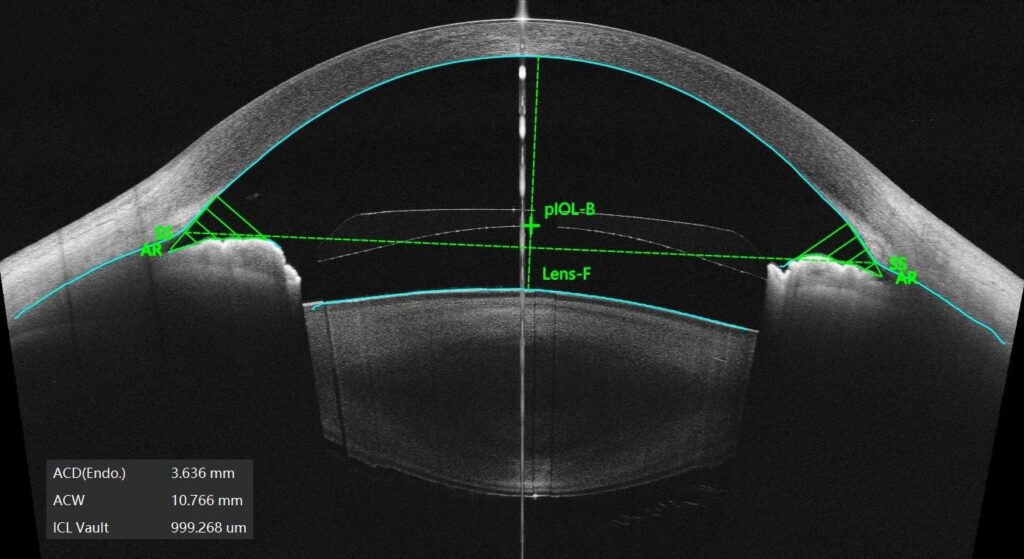
8.1. Background and Rationale
Accurate sizing of Phakic (APL) lenses requires detailed understanding of the spatial anatomical relationships within the anterior segment of the eye. Traditional measurement techniques rely primarily on linear parameters such as White-to-White (WTW) and Anterior Chamber Depth (ACD), which provide indirect estimation of internal anatomical structures. However, Phakic (APL) vault behaviour is influenced by three-dimensional anatomical characteristics including sulcus morphology, iris configuration, crystalline lens position, and anterior chamber geometry.
Three-dimensional (3D) anterior segment modelling enables volumetric reconstruction of the anterior segment, allowing comprehensive visualization of anatomical relationships between the cornea, iris, crystalline lens, anterior chamber angle, and ciliary sulcus. Unlike single-plane cross-sectional imaging, 3D modelling provides spatial representation of the anatomical environment in which the phakic lens is positioned.
Understanding the three-dimensional configuration of the anterior segment is important because vault behaviour depends on spatial compatibility between lens geometry and ocular anatomy rather than a single linear measurement.
8.1.1 Imaging Technology Used for 3D Reconstruction
3D anterior segment modelling is typically performed using high-resolution imaging technologies capable of acquiring multiple cross-sectional scans across different meridians of the anterior segment.
Common imaging modalities include:
These technologies acquire sequential two-dimensional cross-sectional images of the anterior segment at multiple angles. Software algorithms then reconstruct volumetric datasets that represent the three-dimensional geometry of the anterior segment.
Examples of currently available clinical platforms capable of multi-meridian imaging include:
MS-39 anterior segment OCT
Casia swept-source OCT
Anterion OCT imaging platform
Pentacam AXL tomography system
These devices provide high-resolution imaging of anatomical structures relevant to Phakic (APL) sizing.
8.1.2. Method of 3D Anatomical Reconstruction
The process of generating a 3D anterior segment model involves multiple sequential steps:
Step 1 – Image acquisition
Multiple radial cross-sectional scans of the anterior segment are obtained across different meridians using OCT or Scheimpflug imaging.
Step 2 – Landmark identification
Key anatomical landmarks including scleral spurs, anterior chamber angle structures, corneal boundaries, and crystalline lens surfaces are automatically detected using image processing algorithms.
Step 3 – Digital reconstruction
Software reconstructs a volumetric model by integrating multiple cross-sectional images into a continuous anatomical structure.
Step 4 – Geometric analysis
Quantitative measurements of anterior segment geometry are obtained, including spatial relationships between structures influencing Phakic (APL) positioning.
Step 5 – Predictive modelling
Advanced software may simulate lens positioning and estimate postoperative vault distribution based on anatomical configuration.
This process enables evaluation of anatomical relationships in multiple meridians rather than relying on a single cross-sectional measurement.
8.2. Anatomical Parameters Evaluated Using 3D Modelling
Three-dimensional modelling enables evaluation of anatomical characteristics that influence Phakic (APL) sizing but may not be fully captured using conventional linear measurements.
Parameters that can be assessed include:
3D analysis also allows assessment of whether anatomical dimensions are symmetric or asymmetric across different orientations.
Such information is particularly useful when evaluating eyes demonstrating variability in vault outcomes despite similar conventional biometric parameters.
8.3. Relationship Between 3D Anatomy and Vault Behaviour
Vault represents the spatial distance between the posterior surface of the Phakic (APL) lens and the anterior surface of the crystalline lens. Because both structures exhibit curvature and may demonstrate positional variation, vault is influenced by three-dimensional anatomical compatibility.
Factors influencing vault behaviour include:
Eyes with similar WTW values may demonstrate different vault outcomes due to differences in internal anatomical configuration that are better visualized using three-dimensional modelling.
3D modelling helps explain variability in vault distribution across different regions of the lens, including central and peripheral vault differences.
8.4. Role of Artificial Intelligence in 3D Anatomical Analysis
Artificial intelligence algorithms can be integrated with 3D anterior segment modelling to improve prediction of Phakic (APL) sizing outcomes. Machine learning models analyze large datasets of anatomical measurements and postoperative vault outcomes to identify relationships between anatomical geometry and lens behaviour.
AI-based analysis enables:
Advanced analytical tools may simulate multiple lens-size options within the reconstructed anatomical model and estimate the expected vault range.
Such predictive capability supports personalised selection of Phakic (APL) size based on patient-specific anatomical characteristics.
8.5. Comparison with Conventional Linear Measurement Approach
Traditional sizing methods rely primarily on external anatomical measurements that approximate internal ocular dimensions. These methods provide limited information regarding three-dimensional anatomical variability.
3D modelling enables improved anatomical representation of the spatial environment influencing Phakic (APL) positioning.
Comparison of approaches:
| Parameter | Conventional method | 3D modelling approach |
|---|---|---|
| anatomical representation | linear measurement | volumetric assessment |
| sulcus estimation | indirect | anatomical visualization |
| vault prediction | moderate predictability | improved predictability |
| detection of asymmetry | limited | multi-meridian evaluation |
| assessment of tilt | not possible | detectable |
| individual customization | limited | patient-specific |
Three-dimensional modelling provides additional anatomical insight that may improve prediction accuracy in anatomically complex cases.
8.6. Clinical Relevance in Phakic (APL) Sizing
3D anterior segment modelling is particularly valuable in clinical scenarios where conventional measurements provide limited predictive accuracy.
Examples include:
3D anatomical assessment provides additional structural information that may assist in refining lens size selection.
8.9. Current Clinical Availability and Practical Considerations
Although full real-time simulation of Phakic (APL) lens positioning is still evolving, currently available OCT and Scheimpflug imaging platforms provide partial three-dimensional reconstruction of anterior segment anatomy.
These systems are increasingly used in advanced refractive surgery centres to supplement conventional biometric measurements.
Practical considerations include:
Despite these limitations, the integration of three-dimensional anatomical analysis into clinical workflow is expanding.
9. Summary of Methodological Importance
Three-dimensional anterior segment modelling represents an important advancement in anatomical evaluation for Phakic (APL) sizing.
By enabling volumetric assessment of anterior segment geometry, 3D modelling improves understanding of spatial relationships influencing vault behaviour.
Integration of volumetric imaging with artificial intelligence-based predictive tools may contribute to improved predictability of lens sizing outcomes and supports development of individualized anatomical sizing strategies.
Future advancements in computational imaging may further enhance the precision of personalised Phakic (APL) sizing approaches.
Acknowledgement: All figures are schematic educational representations created or adapted from publicly available scientific concepts for explanatory purposes only. No copyright claim is made over original clinical device images.
10. Conclusion
Accurate selection of Phakic (APL) size in mm remains one of the most critical determinants of achieving optimal postoperative vault, long-term safety, and stable refractive outcomes in posterior chamber phakic lens implantation. Traditional sizing methods based primarily on White-to-White (WTW) and Anterior Chamber Depth (ACD) provide a practical and widely accessible starting point; however, these parameters represent indirect anatomical estimations and may not fully reflect the true internal geometry of the eye where the phakic lens is positioned.
Modern anatomical parameters such as Angle-to-Angle (ATA), Sulcus-to-Sulcus (STS), and Lens Rise provide improved understanding of the internal spatial relationships between the cornea, iris, crystalline lens, and ciliary sulcus. These parameters offer enhanced anatomical relevance because they more closely correspond to the actual location of haptic support and the effective internal space available for vault formation. Variations in sulcus morphology, lens position, and anterior chamber configuration explain why eyes with similar WTW measurements may demonstrate different postoperative vault outcomes.
Advances in imaging technologies, including Anterior Segment OCT, Ultrasound Biomicroscopy (UBM), Scheimpflug tomography, and optical biometry, have significantly improved the ability to visualize and quantify anterior segment anatomy. These modalities enable more reliable identification of anatomical variations that influence lens sizing decisions. The integration of Artificial Intelligence (AI) and multivariable prediction models further enhances the ability to analyse complex relationships between anatomical parameters and postoperative vault behaviour, supporting a transition from empirical estimation toward personalized sizing strategies.
Emerging technologies such as 3D anterior segment modelling provide additional insight into spatial anatomical relationships by enabling volumetric reconstruction of the anterior chamber environment. This approach allows improved visualization of lens position, tilt, vault distribution, and anatomical symmetry between eyes. Such advanced modelling techniques contribute to improved understanding of inter-eye variability and may help refine future nomograms for Phakic (APL) sizing.
Based on current evidence, optimal Phakic (APL) size selection should not rely on a single measurement parameter but should incorporate a multivariable anatomical approach, combining traditional measurements with modern imaging-derived parameters. The combined interpretation of WTW, ACD, ATA, STS, lens rise, and anterior chamber configuration provides a more comprehensive understanding of internal ocular anatomy and improves predictability of vault outcomes.
Continued advancements in imaging resolution, computational modelling, and AI-based prediction systems are expected to further improve the precision and reproducibility of Phakic (APL) sizing. Adoption of integrated anatomical assessment protocols may contribute to enhanced surgical safety, improved refractive predictability, and greater consistency in clinical outcomes.
Overall, the evolution from traditional estimation methods toward advanced anatomical and data-driven approaches represents an important step toward personalized refractive surgery planning and improved long-term performance of Phakic (APL) lenses.
Author:
Sudhanshu Sharma, Marketing Manager, Adaptive Ocular Sciences Pvt Ltd., India
MBA, EMBA (IIM)
Email: Sudhanshu.adaptive@gmail.com
11. Fundamental Phakic IOL sizing and vault studies
Role of WTW, ACD and traditional sizing parameters
Angle-to-Angle, Sulcus-to-Sulcus and Lens Rise concepts
Role of OCT and imaging technologies
Scheimpflug imaging and anterior segment tomography
Artificial intelligence in IOL sizing and prediction
3D anterior segment modelling and advanced imaging
This article is intended as an educational review that compiles currently available scientific knowledge, anatomical concepts, and diagnostic considerations related to Phakic (APL) sizing. The content is presented as a structured collection of guiding factors derived from published literature, clinical understanding, and commonly accepted ophthalmic principles.
The material provided in this document is not intended to serve as a prescriptive clinical recommendation, treatment protocol, or mandatory guideline for refractive surgery. The information is designed solely to support medical practitioners, ophthalmologists, and eye surgeons in understanding the anatomical parameters, measurement technologies, and evolving analytical approaches that may influence Phakic (APL) size selection.
Clinical decision-making in refractive surgery involves multiple patient-specific factors, including but not limited to individual anatomical variability, clinical findings, diagnostic interpretation, surgical experience, and professional judgment. Therefore, the final decision regarding suitability, lens sizing, surgical technique, and patient management remains exclusively the responsibility of the treating eye surgeon, who acts as the ultimate authority (apex decision-maker) in determining the most appropriate course of action for each individual patient.
This document should be interpreted as an academic and educational resource intended to facilitate scientific understanding and discussion among qualified healthcare professionals. It does not replace clinical judgment, institutional protocols, regulatory guidance, or individualised patient assessment.
The authors and publishers do not assume responsibility for clinical outcomes resulting from the application or interpretation of the information contained herein. All clinical decisions should be made by appropriately qualified ophthalmic surgeons based on comprehensive evaluation of each patient and in accordance with accepted standards of medical practice.
Image Source Acknowledgement (Short)
Images and diagrams used in this article are included only for educational and scientific illustration purposes. The figures are schematic or concept-based representations derived from commonly available scientific and anatomical references related to anterior segment imaging and Phakic (APL) sizing.
We acknowledge and thank the sources and the ophthalmic scientific community whose published concepts have supported the understanding of:
anterior segment anatomy
OCT and UBM imaging principles
vault concept
3D anterior segment modelling
All trademarks and imaging technologies mentioned belong to their respective owners. No proprietary clinical images or confidential patient data have been used.
These illustrations are intended solely to support learning and scientific communication.