Preoperative Evaluation Framework for Phakic (APL) Implantation
Successful implantation of a Phakic (APL) lens depends on a well-organized and methodical preoperative assessment designed to optimise visual outcomes while maintaining long-term ocular health. Because phakic lenses are placed within the eye rather than on the corneal surface, detailed evaluation of corneal integrity, anterior segment configuration, optical characteristics, and biometric relationships is essential before determining the most appropriate lens parameters.
A. Key Inputs for Phakic IOL Power Determination
Calculation of phakic IOL power is typically based on vergence principles using established mathematical models such as the van der Heijde or Holladay approaches, as well as proprietary formula variations provided by manufacturers. These methods aim to achieve the targeted postoperative refractive state by incorporating the expected effective lens position (ELP), which significantly influences the final optical result. Accurate input data helps improve refractive predictability and reduces the likelihood of residual error.
Key Input Parameters:
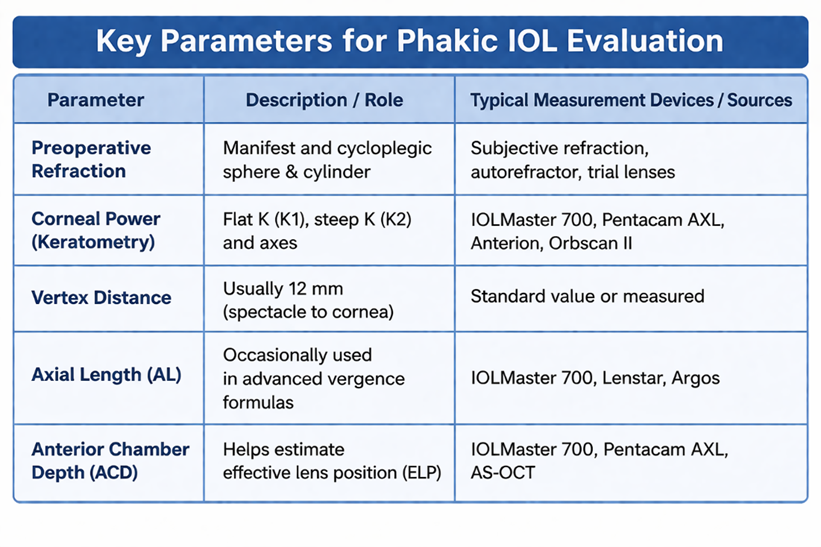
B. Key Inputs for Phakic IOL Size (Overall Diameter) Selection
Selecting the correct lens diameter is fundamental to achieving an appropriate postoperative vault, defined as the distance between the posterior surface of the implanted lens and the anterior capsule of the natural crystalline lens. An optimal vault generally falls within the range of 250–750 µm (0.25–0.75 mm).
• Low vault may increase the risk of anterior subcapsular cataract formation or pupillary block.
• Excessive vault may contribute to narrowing of the anterior chamber angle and potential intraocular pressure elevation.
Therefore, precise anatomical measurement and interpretation are critical for balancing safety with long-term performance.
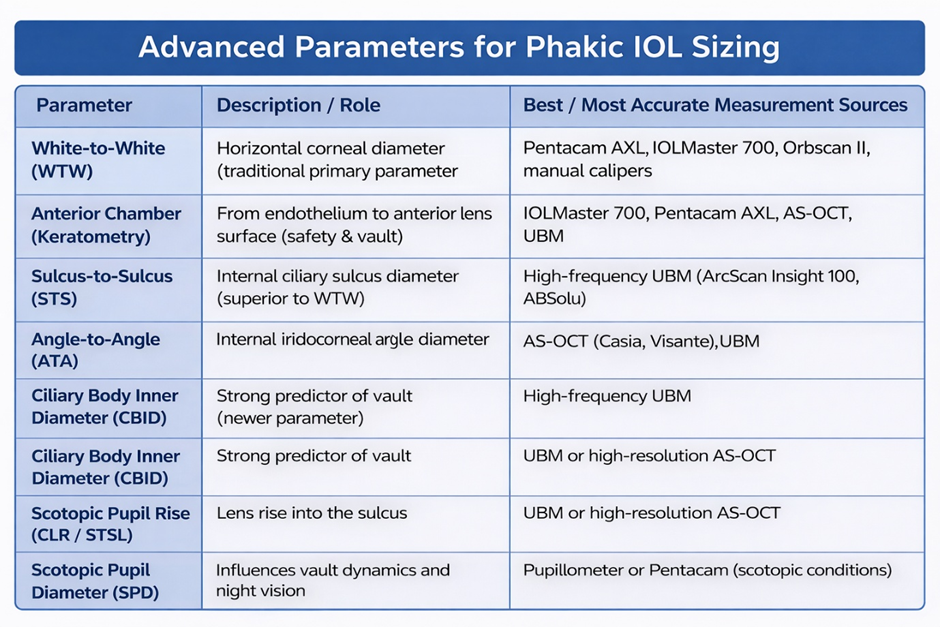
Integrated Diagnostic Workflow in Contemporary Practice
Modern refractive evaluation follows a structured, multi-modal diagnostic pathway that combines information from corneal topography, tomography, wavefront analysis, and ocular biometry. This integrated approach helps reduce the probability of complications such as corneal biomechanical instability, inappropriate vault, higher-order aberrations, or anterior chamber crowding.
Each diagnostic modality contributes unique information regarding ocular structure and optical behaviour. Clinical interpretation requires consideration of both quantitative measurements and qualitative colour-map patterns, ensuring that no single parameter is evaluated in isolation.
The workflow outlined below reflects commonly applied clinical reasoning used by refractive surgeons when determining patient suitability for Phakic (APL) implantation and when aiming to improve consistency and predictability of postoperative visual outcomes.
Preoperative Clinical Decision Pathway for Refractive Surgeons
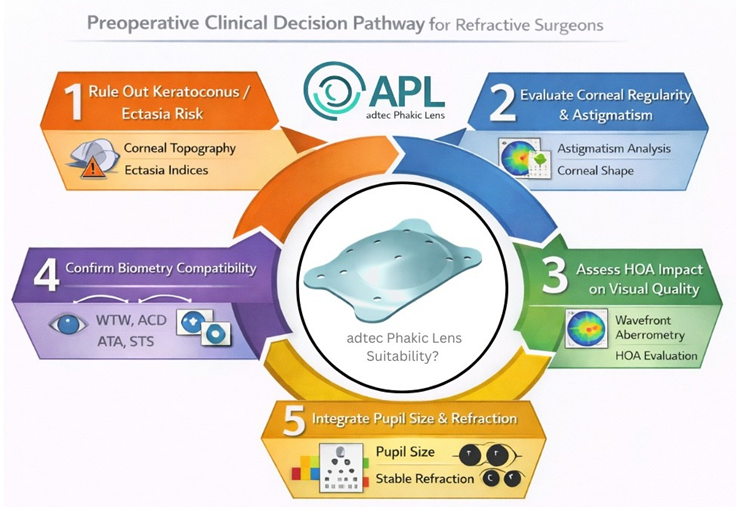
Modern refractive practice follows a stepwise clinical workflow that integrates corneal topography,
tomography, wavefront analysis, and ocular biometry to minimise the risk of postoperative
complications such as ectasia, suboptimal vault, optical aberrations, or angle crowding. Each
diagnostic parameter provides complementary information, and interpretation must consider both
numerical values and colour map patterns in combination.
The following workflow summarises the practical clinical decision logic commonly used by
refractive surgeons to evaluate patient suitability for Phakic (APL) implantation and to improve
predictability of postoperative outcomes.
1️⃣ Rule out keratoconus/ectasia risk
2️⃣ Evaluate corneal regularity & astigmatism
3️⃣ Assess HOA impact on visual quality
4️⃣ Confirm biometry compatibility (WTW, ACD)
5️⃣ Integrate with pupil size & refraction
1️⃣ Rule out Keratoconus / Ectasia Risk
1. Introduction
The initial and most essential stage in screening candidates for Phakic (APL) implantation is the identification and exclusion of corneal ectatic disorders such as keratoconus. Even though phakic lenses do not alter the corneal tissue, the presence of ectasia suggests compromised biomechanical stability and irregular optical quality, which may lead to unpredictable refractive outcomes. Therefore, detailed assessment using corneal topography and tomography is required to confirm that the cornea demonstrates structural integrity before proceeding with lens selection and sizing.
In Phakic (APL) procedures, the corneal shape remains unchanged; however, the cornea continues to serve as the primary refractive surface of the eye. A smooth, regular corneal curvature contributes significantly to visual clarity and refractive predictability after implantation. Conversely, irregular curvature patterns may increase the likelihood of postoperative symptoms such as glare, halos, or reduced contrast sensitivity.
1.1 Axial Curvature Map (Sagittal Map)
The Axial Curvature Map, commonly referred to as the Sagittal Map, represents one of the primary tools used in corneal topography assessment for Phakic (APL) suitability. This map displays the distribution of corneal refractive power using a colour-coded scale that corresponds to keratometry values across the corneal surface.
The sagittal map allows clinicians to evaluate:
Because of its ability to reveal general curvature patterns, the axial map is widely used as the first screening reference to identify forme fruste keratoconus, irregular astigmatism, or asymmetric steepening that may reflect biomechanical weakness.
1.1.1. Evaluation of Corneal Power (K values)
Axial map provides key keratometry parameters:

1.1.1 Assessment of Corneal Power (K Values)
The axial map provides simulated keratometry readings that describe corneal curvature strength.
Typical reference ranges for Phakic (APL) candidates:
Unusually steep corneas may indicate:
Such findings require careful correlation with additional tomography parameters.
1.1.2 Astigmatism Magnitude and Axis (Toric Lens Planning)
The sagittal curvature pattern helps determine whether astigmatic correction with a toric Phakic (APL) lens is appropriate.
Clinical indicators supporting toric correction include:
Situations requiring caution include:
Typical threshold suggesting Toric consideration:
Stable astigmatic patterns generally allow more predictable postoperative refractive outcomes.
1.1.3 Corneal Symmetry as an Indicator of Optical Quality
Corneal symmetry plays an important role in determining expected visual quality following implantation.
Features commonly associated with a normal optical profile: symmetrical bow-tie configuration
Patterns that may indicate irregular optical behaviour include:
Irregular corneal optics may contribute to:
1.1.4 Identification of Early Keratoconus Patterns
One of the important diagnostic functions of the axial curvature map is the early recognition of ectatic changes. Subtle localized steepening, asymmetry between superior and inferior cornea, or distortion of the bow-tie pattern may represent early biomechanical instability. Early detection allows clinicians to avoid inappropriate surgical planning and helps maintain long-term corneal health.
1.1.5 Suspicious Indicators on Axial Curvature Map
Certain curvature patterns on the sagittal map may suggest possible corneal instability and therefore require additional evaluation before confirming suitability for Phakic (APL) implantation.
Common warning signs include:
When such findings are present, interpretation should not rely on the axial map alone. Correlation with additional diagnostic indices improves accuracy and reduces the risk of overlooking early ectatic change.
Further evaluation typically includes:
Integrated assessment improves confidence in identifying subtle biomechanical abnormalities that may influence refractive predictability.
1.1.6 Relationship Between Corneal Optics and Final Visual Quality
Visual performance after Phakic (APL) implantation depends on the combined optical contribution of the cornea and the implanted intraocular lens.
Final optical performance is influenced by:
Corneal optics + intraocular lens optics
If the corneal surface demonstrates irregular curvature or higher-order aberrations, optimal visual quality may not be achieved even when lens power selection is accurate. Therefore, evaluation of corneal regularity plays a significant role in predicting postoperative clarity, contrast sensitivity, and night vision quality.
1.1.6 Quick Clinical Interpretation Guide
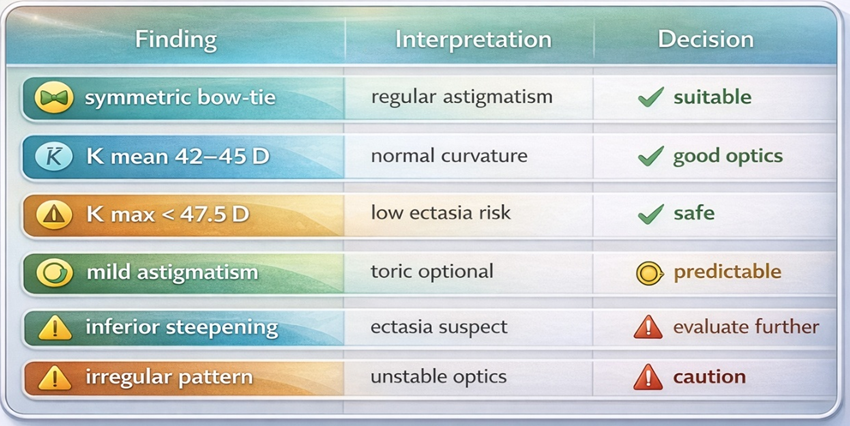
Clinical Importance in Phakic (APL) Planning
1.1.7 Clinical Importance of Axial Curvature Map in Phakic (APL) Planning
Interpretation of the sagittal curvature map provides essential preliminary information that supports clinical decision-making.
Key roles include:
For these reasons, sagittal map analysis represents a fundamental first step in the screening workflow.
1.2 Pachymetry Map (Corneal Thickness Distribution)
Introduction
The pachymetry map illustrates the spatial distribution of corneal thickness using a colour-coded display. This parameter is particularly relevant in Phakic (APL) assessment because corneal thickness indirectly reflects biomechanical integrity and structural uniformity.
In physiologically normal corneas, thickness gradually increases from the central region toward the periphery, producing a smooth and symmetrical distribution pattern. Evaluation of thickness characteristics provides information that complements curvature analysis, allowing identification of structural abnormalities that may not be apparent on topography alone.
Irregular thickness patterns may suggest early ectatic change, including subclinical keratoconus, which could affect long-term optical stability. Therefore, pachymetry analysis contributes to safe candidate selection by confirming structural consistency of the cornea.

1.2.2 Normal Pachymetry Characteristics
A typical pachymetry profile demonstrates the thinnest region near the central cornea, with progressive thickening toward peripheral zones.
Features commonly associated with normal thickness distribution include:
Such findings suggest normal anatomical architecture and adequate biomechanical stability.
1.2.3 Abnormal Pachymetry Patterns
Deviation from the expected thickness profile may indicate structural variation requiring further evaluation.
Examples of atypical pachymetry findings include:
These variations may represent early biomechanical alteration, even when curvature patterns appear relatively normal. Careful interpretation helps reduce the risk of overlooking subtle ectatic changes.
1.2.4 Assessment of the Thinnest Corneal Location
Evaluation of the thinnest corneal point provides valuable insight into structural symmetry.
In normal corneas:
Significant displacement of the thinnest point, particularly toward inferior or inferotemporal regions, may indicate localized structural asymmetry.
Analysis of this parameter supports early detection of biomechanical variation.
1.2.5 Thickness Progression Pattern
Assessment of how corneal thickness changes from the centre toward the periphery helps determine whether the structural profile follows a physiologically normal pattern.
Normal pattern:
Abnormal pattern:
Such irregularities may reflect localized biomechanical weakening or altered structural organization and therefore require careful clinical interpretation.

1.2.6 Interpretation Principles
Pachymetry maps should be assessed systematically by evaluating:
A structured interpretation approach improves detection of subtle structural variation and enhances diagnostic reliability.
1.2.7 Clinical Importance in Phakic (APL) Evaluation
The pachymetry map contributes to the assessment of corneal structural stability by identifying thickness distribution characteristics that may indicate early ectatic change. Abnormal thickness patterns, localised thinning, or displacement of the thinnest point may suggest the need for careful evaluation before proceeding with refractive correction.
For comprehensive screening, pachymetry findings should be interpreted together with axial curvature maps, posterior elevation maps, and BAD-D index values. Combined assessment of these parameters improves the sensitivity of ectasia detection and supports reliable clinical decision-making in Phakic (APL) selection.
1.2.8 Colour Code Interpretation (Thickness Map)

Important note:
The thickness colour scale is the opposite of the curvature map interpretation.
1.2.9 Belin–Ambrosio Enhanced Ectasia Display (BAD-D)
The Belin–Ambrosio Enhanced Ectasia Display (BAD-D) is one of the most reliable and widely used indices for detecting early keratoconus and corneal ectasia risk. It combines multiple tomography parameters into a single deviation value (D value), allowing clinicians to identify subtle structural abnormalities even when individual maps appear normal.
For Phakic (APL) screening, BAD-D plays an important role in confirming corneal biomechanical stability, ensuring that the cornea does not show early ectatic changes that may affect long-term visual quality. Since Phakic lenses provide high optical precision, underlying corneal irregularities may reduce expected visual outcomes.
BAD-D integrates both elevation-based and pachymetry-based parameters, making it more sensitive than single-map evaluation.
Components of BAD-D Index
BAD-D is derived from five deviation parameters:
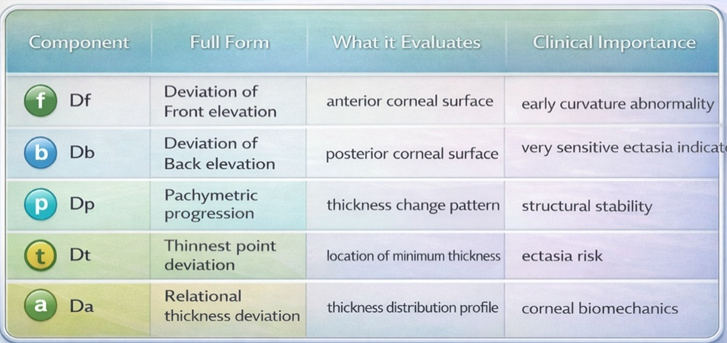
The final BAD-D value combines all deviations into a single index.
BAD-D Interpretation Guide

2️⃣ Evaluate Corneal Regularity & Astigmatism
Evaluation of corneal regularity and astigmatism is an essential step in Phakic (APL) planning, as the implanted lens corrects refractive error but does not change corneal shape. Therefore, the quality of corneal optics directly influences the final visual outcome.
A regular and symmetrical corneal surface allows accurate correction of sphere and cylinder power, while irregular corneal patterns may result in residual refractive error, glare, halos, reduced contrast sensitivity, and patient dissatisfaction.
Proper interpretation of keratometry values (K1, K2), astigmatism magnitude, axis stability, and symmetry pattern helps determine whether toric or non-toric Phakic (APL) should be selected.
2.1. Axial Curvature Pattern (Regular vs Irregular Astigmatism)
Phakic lenses provide excellent optical correction when the corneal surface is regular and stable.
Regular astigmatism produces predictable visual correction with toric Phakic IOL, while irregular astigmatism may cause poor quality of vision even if refractive error is corrected.

2.2 Keratometry Values (K1, K2, K Mean, K Max)
Keratometry indicates corneal refractive power and helps confirm optical regularity.
Important cut-off values for Phakic IOL

A very steep or irregular cornea may indicate:
Phakic IOL works best when corneal optics are stable.
2.3. Astigmatism Magnitude (Cylinder Power)
Cylinder magnitude helps determine whether a toric Phakic IOL is required.
Clinical cut-off values

2.4. Axis Stability (Repeatability of Measurement)
The axis of astigmatism should be consistent between:
manifest refraction
topography
tomography Acceptable variation

Large axis variation indicates unstable or irregular astigmatism.
2.5. Corneal Symmetry (Inter-eye Comparison)
Assessment of corneal symmetry between both eyes is an important screening parameter in Phakic (APL) planning, as the human cornea typically demonstrates a high degree of bilateral similarity in curvature pattern, astigmatism magnitude, and optical quality. Significant asymmetry between the right and left eye may indicate subclinical corneal pathology, early keratoconus (forme fruste keratoconus), irregular astigmatism, or measurement inconsistency, all of which can affect refractive predictability and postoperative visual performance.
Since Phakic (APL) provides internal optical correction without modifying corneal structure, the final visual outcome depends strongly on the pre-existing corneal optical symmetry and regularity. Therefore, comparison of keratometry values, astigmatism magnitude, and axis orientation between both eyes helps confirm corneal stability and the reliability of measurements.
Inter-eye comparison is particularly useful when individual eye parameters fall within normal limits, but subtle asymmetry suggests early biomechanical instability.
Both eyes should show a similar corneal pattern.
Acceptable inter-eye variation

Large asymmetry may indicate:
3️⃣ Assess HOA Impact on Visual Quality
Higher Order Aberrations (HOA) represent subtle optical irregularities of the eye that cannot be corrected with simple sphere or cylinder lenses. Even when refractive error is fully corrected with Phakic (APL), increased HOA may reduce the quality of vision, causing symptoms such as glare, halos, starburst, ghost images, and reduced contrast sensitivity, especially under low-light (scotopic) conditions.
Since Phakic IOL provides high-quality internal optics, the final visual outcome largely depends on pre-existing corneal optical quality. Therefore, evaluation of HOA is important to ensure that implantation results in sharp and stable visual performance.
HOA assessment is typically obtained from wavefront Aberrometry, corneal topography, or tomography systems such as Pentacam, iTrace, or ray-tracing aberrometers.
3.1 HOA Map Interpretation
HOA maps show the distribution of optical irregularities using colour-coded patterns.
Smooth and symmetric pattern → better optical quality
Irregular scattered pattern → reduced visual quality
3.2 Important HOA Parameters for Phakic IOL
A. Total HOA RMS (Root Mean Square)
Indicates the overall magnitude of higher-order aberrations.
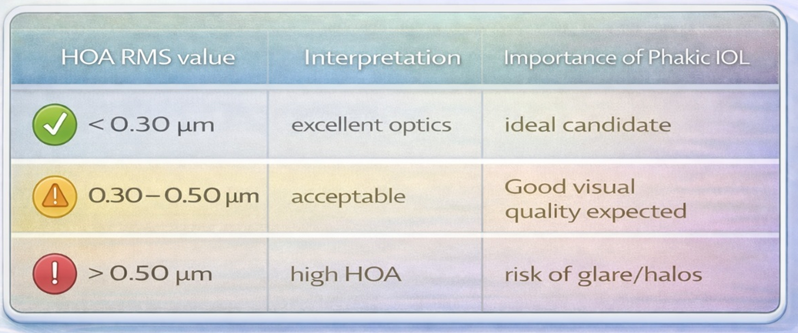
Lower HOA values provide:
B . Coma Aberration
Coma is one of the most clinically significant aberrations, often associated with irregular corneal shape or early keratoconus.

High coma aberration is clinically significant because it produces asymmetrical distortion of the retinal image. Patients with elevated coma values may experience visual symptoms such as ghost images, comet-shaped light distortion, and reduced clarity of objects, particularly under low illumination conditions. Coma is commonly associated with irregular corneal shape or early biomechanical instability and may negatively influence night vision quality even when refractive correction is accurate.
Spherical aberration represents variation in the focusing of peripheral light rays compared with central rays entering the eye. When spherical aberration is elevated, peripheral rays focus at a different point than central rays, resulting in optical blur. Increased spherical aberration may cause visual disturbances such as glare, halos around lights, and reduced contrast sensitivity, particularly in dim lighting, when pupil size increases. Phakic (APL) optics perform optimally when spherical aberration remains within physiological limits, as this allows more uniform focusing of light and improved retinal image quality.
D. Trefoil Aberration
Trefoil represents asymmetric optical distortion.
Higher values may lead to:
Typical Acceptable HOA Range for Phakic IOL

Importance of HOA in Phakic IOL Planning
Core Concept
Visual Symptoms Associated with High HOA
Clinically Acceptable Cut-off Values

Why HOA Evaluation is Important for Phakic (APL)
HOA assessment helps to:
Parameters to Correlate with HOA
HOA should always be interpreted along with:
Combined evaluation improves the reliability of screening and helps ensure optimal visual quality after Phakic (APL).
4️⃣ Confirm Biometry Compatibility (WTW, ACD)
Biometric compatibility is one of the most critical steps in Phakic (APL) sizing, as the lens is positioned inside the eye between the iris and crystalline lens. Proper matching of lens size with internal ocular anatomy helps achieve an optimal vault, ensuring long-term safety and stable visual performance.
Among all biometric parameters, White-to-White (WTW) and Anterior Chamber Depth (ACD) are the most commonly used measurements in manufacturer calculators. These parameters provide an estimation of the available anatomical space for proper positioning of the phakic lens.
Incorrect interpretation of these values may lead to:
low vault → risk of anterior subcapsular cataract
high vault → angle crowding and increased IOP
Therefore, accurate measurement and cross-verification are essential.
Key Biometric Parameters
4.1 White-to-White (WTW)
WTW represents the horizontal visible corneal diameter measured from limbus to limbus.
Importance of Phakic IOL sizing
WTW is traditionally used to estimate the internal anatomical size of the eye and select the overall lens length.
4.2 Typical acceptable range

WTW alone does not represent internal sulcus size but provides a useful starting point when combined with ACD.
4.3 Anterior Chamber Depth (ACD)
ACD measures the distance from the corneal endothelium to the anterior crystalline lens surface.
ACD is critical for ensuring sufficient space for safe implantation.
Typical cut-off values for Phakic (APL)

A reduced anterior chamber depth (ACD) is an important consideration during Phakic (APL) planning because the lens is positioned within the anterior segment. Limited intraocular space may influence lens positioning and postoperative fluid dynamics.
4.4 Relationship of WTW, ACD, ATA, STS and Lens Rise with Postoperative Vault
4.4.1 Introduction
Accurate prediction of postoperative vault is a key objective in Phakic (APL) sizing, as vault represents the distance between the posterior surface of the phakic lens and the anterior surface of the crystalline lens. Appropriate vault is important for maintaining physiological separation between these structures and supporting long-term safety.
Traditionally, White-to-White (WTW) and Anterior Chamber Depth (ACD) have been used as primary parameters for lens sizing due to their accessibility across common diagnostic devices. However, these measurements represent indirect anatomical indicators and may not fully reflect the internal geometry of the ciliary sulcus where the phakic lens is positioned. Consequently, additional anatomical parameters such as Angle-to-Angle (ATA), Sulcus-to-Sulcus (STS), and Lens Rise are increasingly considered to improve vault predictability and reduce variability between eyes.
4.4.2 Role of White-to-White (WTW)
WTW represents the horizontal visible corneal diameter measured between the limbal boundaries. Because of its simplicity and availability across multiple imaging platforms, WTW has historically been incorporated into most phakic lens sizing nomograms.
In clinical practice, larger WTW measurements are generally associated with selection of longer lens sizes, which may contribute to higher postoperative vault, while smaller WTW values may correspond to shorter lens sizes and relatively lower vault. However, WTW reflects external corneal anatomy and does not directly measure the internal sulcus diameter. As a result, reliance on WTW alone may not fully capture anatomical variation relevant to vault prediction, particularly when internal ocular dimensions differ from external corneal measurements.
Typical clinical range:
WTW 11.0 – 12.5 mm is considered the common anatomical range.
Extreme WTW values require confirmation using multiple devices to reduce measurement variability.
4.4.3 Role of Anterior Chamber Depth (ACD)
ACD measures the distance from the corneal endothelium to the anterior surface of the crystalline lens and represents the available anatomical space for lens implantation. A deeper anterior chamber generally allows greater space for the phakic lens and contributes to a more stable vault profile.
General clinical trend indicates that deeper ACD values are associated with more favourable vault stability, whereas shallow anterior chamber depth may increase the risk of excessive anterior displacement of the lens.
Typical clinical interpretation:
ACD should always be interpreted together with angle anatomy and internal anterior segment dimensions.
4.4.4 Role of Angle-to-Angle (ATA)
Angle-to-Angle distance represents the internal horizontal width of the anterior chamber measured between scleral spurs. ATA provides a better representation of internal anterior segment anatomy compared with WTW because it directly reflects the internal anatomical space where the phakic lens haptics interact with the ciliary body.
ATA measurement is typically obtained using anterior segment OCT or Scheimpflug tomography.
Clinical relevance:
ATA is particularly useful when WTW values appear similar, but the postoperative vault differs between eyes.
4.4.5 Role of Sulcus-to-Sulcus (STS)
Sulcus-to-Sulcus distance represents the internal diameter of the ciliary sulcus, which is the actual anatomical location where the phakic lens rests. STS measurement provides a more direct anatomical representation of lens sizing requirements compared with WTW.
STS is typically measured using Ultrasound Biomicroscopy (UBM) or high-resolution anterior segment OCT.
Clinical importance:
Eyes with similar WTW may demonstrate different STS measurements, explaining variability in vault outcomes.
4.4.6 Role of Lens Rise
Lens Rise refers to the anterior protrusion of the crystalline lens relative to the angle-to-angle reference plane. It represents the relative position of the natural lens within the anterior segment and significantly influences the final vault achieved after implantation.
Higher lens rise indicates a greater forward position of the crystalline lens, which may reduce vault due to decreased posterior space available for the phakic lens.
Lower lens rise indicates a more posterior crystalline lens position, which may result in increased vault.
Clinical Significance of Lens Rise in Phakic (APL) Sizing
Clinical importance:
Eyes with similar WTW may demonstrate different STS measurements, explaining variability in vault outcomes.
4.4.6 Role of Lens Rise
Lens Rise refers to the anterior protrusion of the crystalline lens relative to the angle-to-angle reference plane. It represents the relative position of the natural lens within the anterior segment and significantly influences the final vault achieved after implantation.
Higher lens rise indicates a greater forward position of the crystalline lens, which may reduce vault due to decreased posterior space available for the phakic lens.
Lower lens rise indicates a more posterior crystalline lens position, which may result in increased vault.
Clinical Significance of Lens Rise in Phakic (APL) Sizing
Lens rise represents the anterior position of the crystalline lens relative to the horizontal reference line connecting the scleral spurs. It provides additional anatomical insight that is not captured by traditional horizontal white-to-white (WTW) measurement alone.
In modern Phakic (APL) planning, lens rise has become an important parameter because vault behaviour depends not only on horizontal ocular dimensions but also on the anterior-posterior relationship between the crystalline lens and the anterior chamber.
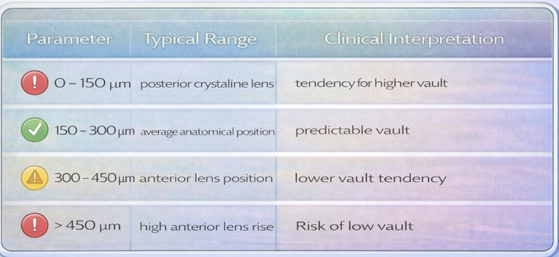
Lens rise is typically measured using AS-OCT or UBM imaging.
4.4.7 Multivariable Relationship with Vault
Postoperative vault is influenced by the interaction of multiple anatomical parameters rather than a single measurement.
General clinical trend:
Therefore, the combined interpretation of external and internal anatomical parameters improves prediction accuracy.
4.4.8 Practical Clinical Interpretation
Suitable anatomical profile generally demonstrates:
Caution is advised when:
ACD less than 2.8 mm
significant inconsistency between measurements
unusual anatomical configuration
4.4.9 Clinical Importance in Phakic (APL) Sizing
Biometric compatibility ensures appropriate lens sizing, optimal vault range, stable lens positioning, and reduced risk of postoperative complications. Although WTW and ACD remain widely used parameters, improved accuracy in phakic lens sizing is achieved when these values are interpreted together with ATA, STS, lens rise, and anterior chamber angle configuration.
Multivariable anatomical assessment improves prediction of postoperative vault, reduces unexpected vault outcomes, and supports development of more personalised phakic lens sizing strategies.
5️⃣ Integrate with Pupil Size & Refraction
Final decision for Phakic (APL) implantation should integrate refractive error characteristics and pupil behaviour, as these factors influence visual quality, night vision symptoms, and lens selection (toric vs non-toric).
Pupil size affects how light enters the eye, particularly in dim illumination. Larger scotopic pupils may increase the risk of glare, halos, or optical disturbances if optical zone coverage is insufficient.
Refraction analysis confirms the magnitude and stability of refractive error before selecting lens power.
5.1 Pupil Size (Photopic, Mesopic and Scotopic Conditions)
Pupil diameter varies according to illumination conditions and plays an important role in determining the optical performance of Phakic (APL), particularly under low-light environments. Since the optical zone of the implanted lens interacts with incoming light rays, larger pupil sizes may expose peripheral optical zones, which can influence visual quality, especially at night.
Pupil size is typically evaluated under three illumination levels:

A larger scotopic pupil allows peripheral light rays to enter the eye, increasing the possibility of optical phenomena such as glare, halos, starburst, and reduced contrast sensitivity. These effects may be more noticeable in patients with high refractive correction because higher optical power increases sensitivity to optical imperfections.
Modern diagnostic devices such as Pentacam, pupillometer, and wavefront aberrometers provide reliable measurement of pupil size under controlled lighting conditions. Evaluation of pupil dynamics helps in predicting night vision quality and patient adaptation after Phakic (APL) implantation.
Clinical relevance:
A large scotopic pupil (>6.5–7 mm) may increase the probability of night vision disturbance
Significant inter-eye pupil asymmetry may require further neurological or pharmacological evaluation
Pupil size should be correlated with the HOA and the optical zone diameter of the phakic lens
Modern lens designs aim to reduce optical aberrations; however, very large pupils may still produce visual symptoms under low illumination.
5.2 Refractive Error (Sphere and Cylinder)
Accurate and stable measurement of refractive error is fundamental for determining the appropriate optical power of the Phakic (APL). Stability of refraction ensures that the refractive error is not undergoing active progression, thereby improving the predictability of postoperative visual outcome.
Refraction assessment should include:
manifest refraction
cycloplegic refraction
correlation with topography and tomography
Typical correction ranges for Phakic (APL):
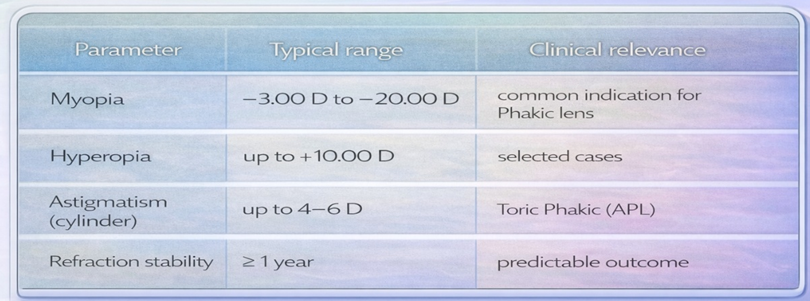
Stable refraction is important because progressive myopia or unstable astigmatism may lead to postoperative residual refractive error.
Cylinder magnitude and axis stability help determine whether toric Phakic (APL) is required. Cylindrical refractive error greater than approximately 0.75–1.00 D is generally considered clinically significant and may benefit from toric correction, provided the axis demonstrates repeatability across multiple measurements.
Higher refractive error often requires careful evaluation of optical quality parameters such as HOA, pupil diameter, and corneal regularity, as these factors influence final visual performance.
Clinical Importance in Phakic (APL) Selection
Evaluation of pupil size and refractive error contributes to:
to ensure a comprehensive and individualised approach to Phakic (APL) planning.
Confirmation of Refraction Before Phakic (APL) Implantation
Accurate confirmation of refractive error is essential before Phakic (APL) implantation to ensure appropriate lens power selection and a predictable visual outcome. Because phakic lenses provide precise optical correction, even small inaccuracies in refraction may result in residual error or reduced visual quality. Therefore, refraction should be confirmed using complementary assessment methods to ensure measurement reliability.
Manifest refraction is a subjective measurement obtained through patient feedback using trial lenses or a phoropter. It reflects the patient’s functional visual requirement and serves as the primary reference for determining spherical and cylindrical components of refractive error, as well as assessing visual acuity potential and acceptance of correction.
Cycloplegic refraction is performed after pharmacological relaxation of accommodation, allowing identification of latent hyperopia or accommodative influence that may alter the true refractive status. This method is particularly useful in younger individuals or patients with active accommodation and helps confirm the stability of spherical power.
Comparison with corneal topography or tomography provides an objective relationship between refractive and corneal astigmatism. Agreement between refractive cylinder axis and corneal steep axis supports measurement accuracy, whereas significant differences may indicate irregular astigmatism, lenticular contribution, tear film variability, or measurement inconsistency that should be reviewed before finalising lens power.
Overall, consistency among manifest refraction, cycloplegic refraction, and corneal imaging findings increases confidence in refractive stability and supports accurate selection of lens power for Phakic (APL) implantation.
Importance of Cylinder Stability in Toric Phakic (APL) Planning (De-duplicated Version)
Toric Phakic (APL) is generally considered when cylindrical refractive error is clinically significant (typically ≥ 0.75–1.00 D) and when the astigmatism axis demonstrates stability across repeated measurements. Accurate cylinder magnitude and consistent axis orientation are necessary for proper toric lens alignment, as rotational deviation or axis variability may reduce astigmatic correction effectiveness and result in residual refractive error.
Consistency between manifest refraction and corneal topography improves confidence in the reliability of astigmatism measurement. Variability in cylinder magnitude or axis may indicate irregular astigmatism, tear film instability, or measurement inconsistency, which can reduce the predictability of toric lens performance.
Clinically favourable outcomes with Toric Phakic (APL) are typically associated with stable refractive findings over time, agreement between refractive and corneal astigmatism, and normal physiological pupil size. Borderline situations may occur when cylindrical error is close to the lower treatment threshold, mild axis variation is observed between measurements, or scotopic pupil size is relatively large (approximately >7 mm). In such cases, careful correlation with corneal regularity and overall optical quality parameters supports appropriate case selection and optimal visual outcome.
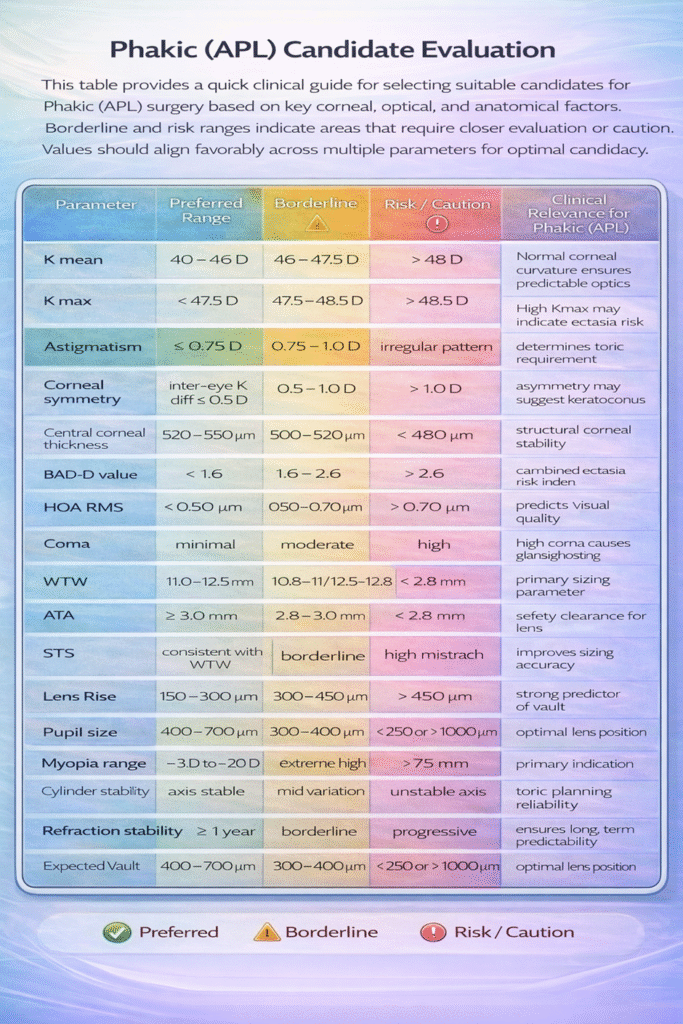
Conclusion
Accurate Phakic (APL) size selection in millimetres requires a multidimensional clinical evaluation rather than reliance on a single parameter. While traditional measurements such as White-to-White (WTW) and Anterior Chamber Depth (ACD) remain fundamental for initial estimation, modern refractive practice increasingly integrates additional anatomical and optical parameters, including Angle-to-Angle (ATA), Sulcus-to-Sulcus (STS), Lens Rise, corneal tomography indices, and Higher Order Aberrations (HOA), to improve predictability of postoperative vault and visual quality.
The combined interpretation of corneal shape, corneal thickness distribution, BAD-D ectasia index, optical aberration profile, and anterior segment anatomy allows clinicians to better identify suitable candidates and reduce the risk of complications such as excessive or inadequate vault, angle crowding, glare, halos, or suboptimal visual quality. Parameters such as HOA RMS (<0.50 µm), K values within normal range, adequate ACD (≥ 3.0 mm), stable refraction, and consistent inter-eye symmetry contribute to predictable surgical outcomes and long-term safety.
Modern imaging modalities, including Scheimpflug tomography, AS-OCT, and UBM, provide improved visualisation of internal ocular anatomy, while emerging AI-based prediction models and 3D anterior segment modelling enhance the understanding of spatial relationships between the phakic lens and surrounding structures. These advances support more individualised lens sizing strategies and help explain inter-eye differences in vault that may not be predictable from WTW alone.
The summary table presented in this article serves as a practical clinical reference to integrate corneal safety screening, optical quality assessment, and anatomical compatibility into a structured decision-making pathway. A systematic approach to evaluating these parameters can improve sizing accuracy, optimise vault outcomes, and enhance patient satisfaction following Phakic (APL) implantation.
Overall, successful Phakic (APL) outcomes depend on a comprehensive interpretation of anatomical, biomechanical, and optical factors together, supporting a shift from traditional single-parameter estimation toward multivariable, evidence-based sizing methodology.
Author:
Sudhanshu Sharma, Marketing Manager, Adaptive Ocular Sciences Pvt Ltd., India
MBA, EMBA (IIM)
Email: Sudhanshu.adaptive@gmail.com
References
Declaration:
This article on Phakic (APL) sizing is intended purely as an educational review, compiling current scientific knowledge, anatomical concepts, and diagnostic considerations relevant to lens size selection. The content represents a structured summary of guiding factors based on published literature and accepted ophthalmic principles.
This material does not constitute a clinical recommendation, treatment protocol, or mandatory guideline. It is designed only to support ophthalmologists and eye surgeons in understanding parameters and technologies that may influence Phakic lens sizing.
Final decisions regarding patient suitability, lens size, and surgical management remain solely the responsibility of the treating eye surgeon, whose clinical judgment is the ultimate authority for each patient.
This material is designed for use by appropriately trained medical practitioners and is provided for educational and academic reference purposes only. It is not intended to replace professional clinical judgement, established institutional guidelines, or individualized patient assessment. Clinical decisions should always be based on the practitioner’s expertise, patient-specific findings, and applicable regulatory and professional standards.